North Texas educators hopeful vaccine could bring some normalcy to upcoming school year

North Texas educators hopeful vaccine could bring some normalcy to upcoming school year
With the news of the Pfizer vaccine being 100% effective among kids between 12 and 15, educators are hoping it will free more students to return to the classroom this fall.
SUNNYVALE, Texas - With the news of the Pfizer vaccine being 100% effective among kids between 12 and 15, educators are hoping it will free more students to return to the classroom this fall.
In Pfizer's clinical trial, none of the 2,200 children got COVID while 18 of those in the placebo group did come down with the virus. It could mean kids as young as 12 could start to get the vaccine before heading back to school in August.
MORE: Pfizer says its COVID-19 vaccine is safe, strongly protective in kids as young as 12
Caleb Chung is one of Pfizer's 2,060 participants in a trial to determine if kids as young as 12 can receive the vaccine. It is currently authorized for those 16 and up.
"It’s potentially helping other kids feel safe and I wanted to get the vaccine as a way I could help out," he said.
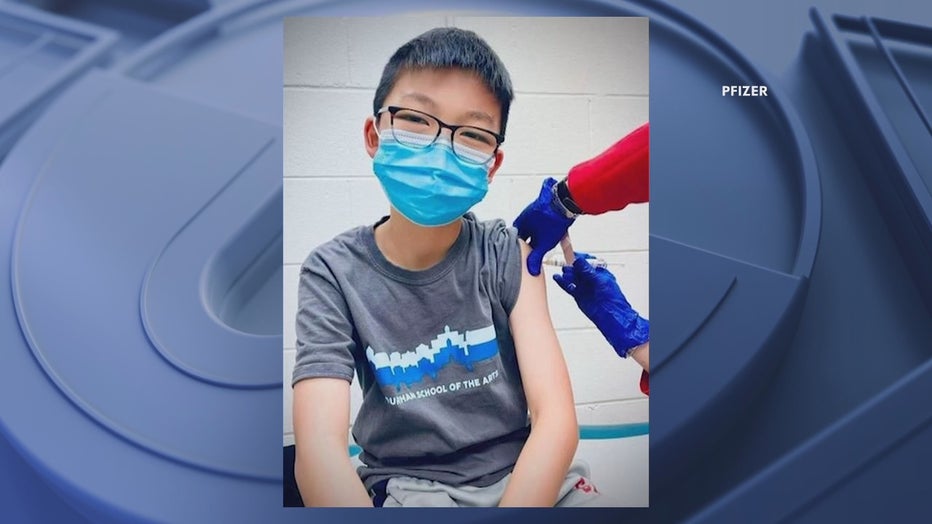
Caleb's dad, Dr. Richard Chung, is a pediatrician at Duke University. He said his son was happy to volunteer, despite the many blood tests the trial required.
"It was special," he said. "I’m proud of him for stepping up in this way, and we are excited. As a pediatrician, vaccinations have always been front of mind. They’re critical to keeping kids safe."
Parkland CEO Dr. Joseph Chang said the results are encouraging.
"At the beginning, there was no reason to think these vaccines would not be safe for younger folks," he said. "It’s good to have proof. It’s very exciting for what the fall looks like for our school."
North Texas superintendents believe time is of the essence for getting kids back in the classroom.
"We firmly believe kids learn best from each other. It’s the best methodology," said Sunnyvale ISD Superintendent Doug Williams. "That's been more difficult to accomplish online."
Nearly 30% of Sunnyvale ISD's students are still virtual.
"Most cases, we see are middle and high school," Williams said. "We think that would reassure more of our parents to be able to send kids back to in person instruction."
Caleb is eager for that opportunity as well.
"I hope we get back to whatever we call normal eventually soon," he said.
In Pfizer's trial, kids had the same side effects as adults from the vaccine, such as pain, fever, chills, and fatigue, particularly after the second dose.
Researchers also found higher levels of virus-fighting antibodies than in their studies of young adults.

