Moderna COVID vaccine trial underway on children in Plano

Moderna COVID vaccine trial underway on children in Plano
Clinical trials to test the Moderna vaccine's effectiveness in children are getting underway in North Texas.
PLANO, Texas - Clinical trials to test the Moderna vaccine's effectiveness in children are getting underway in North Texas.
And while the trial will monitor children for a year, the FDA could grant emergency use authorization much sooner.
12-year-old Leighton Horan hopes to help make history. She received a shot that either contained the Moderna COVID-19 vaccine or a placebo.
"I hope it was the vaccine," she said. "But either way, I know I am helping kids all over the world."
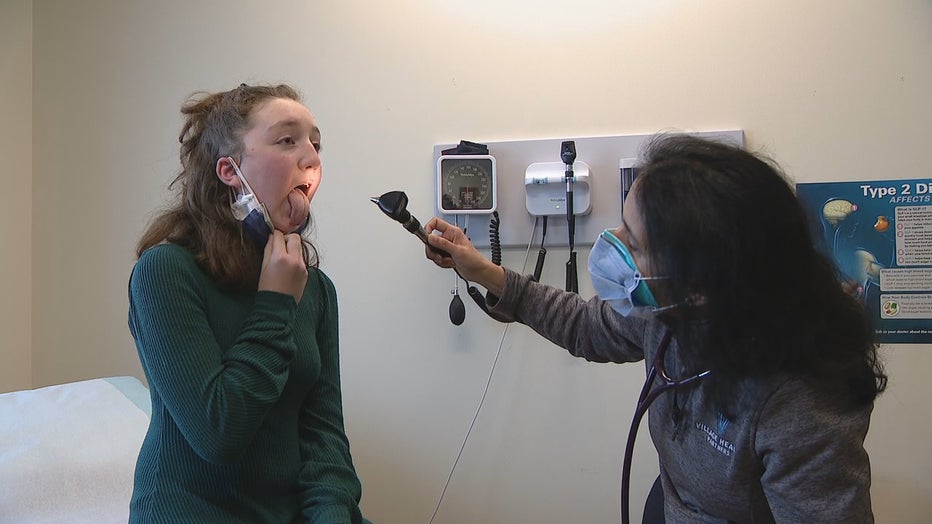
The pandemic turned Leighton's world upside down. She’s been in virtual school for nearly a year now and is missing out on the typical school experiences so many hold dear.
Because this is the Moderna product, it does not use a weakened live virus.
Dr. Madhavi Ampajwala says the technology is better than the old method.
"It teaches the immune system how to react," she said.
The messenger RNA vaccines teach cells how to fight COVID-19 using a manufactured material.
"It’s great science and technology that I’m hoping future vaccines will use," Dr. Ampajwala said. "It has been around, but these vaccines are first in its class."
While earlier trials for adults needed help finding participants ACRC trials has found youth are eager to get involved.
ACRC Trials has a waitlist 1,000 kids long.
"Teens are excited they get to contribute and learn," said ACRC Trials Director Heema Marwah. "I think because they get to talk to friends and wonder how they got vaccinated because it’s not available yet."
By offering up herself as a trial participant, Leighton hopes to help get scientists get the answers they need sooner.
While registration for this trial is closed, ACRC plans to have another clinical trial to study the vaccine in younger children.

